 
Atomic Mass 12,011 Learn more about the atomic mass. All possible symmetric arrangements of particles in three-dimensional space are described by the 230 space groups (219 distinct types, or 230 if chiral copies are considered distinct. The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Question: the atomic radius of carbon is about 70 pm, or about 70 x 10-12 m. Radiocarbon dating uses the carbon-14 isotope to date old objects. The symmetry properties of the crystal are described by the concept of space groups. 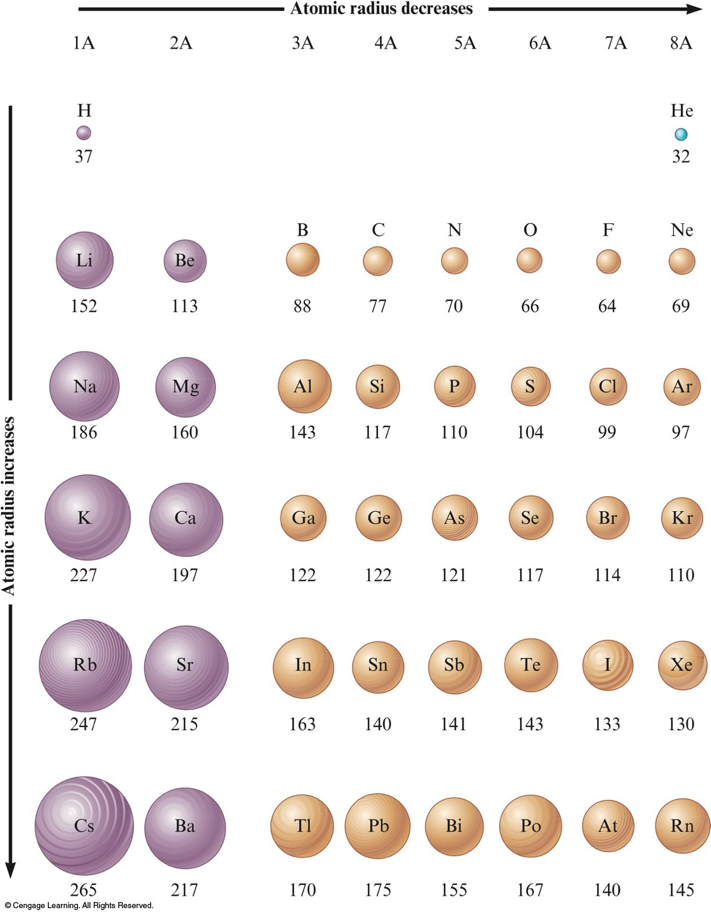
The positions of the atoms inside the unit cell are described by the set of atomic positions ( x i, y i, z i) measured from a reference lattice point. The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges Lattice Constants (a, b and c) aĪnd the angles between them Lattice Angles (alpha, beta and gamma). The unit Cells repeats itself in three dimensional space to form the structure. The Crystal structure can be described in terms of its unit Cell. The solid state structure of Carbon is Simple Hexagonal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
